Biomechanics of Native Heart Valves
The extracellular matrix (ECM) characteristics play a key role in the pathophysiology of native valves. In our lab, we study the ECM organization of native and bioprosthetic heart valve tissues in response to different mechanical loading conditions. This is of critical importance since ECM components and mainly collagen fibers provide structural integrity withstanding the cyclic loads during systole and diastole, and compromise in collagen structure and function can determine the pathological fate of the valves. We have developed a novel loading/imaging technique that takes advantage of a custom made biaxial mechanical loading device that can be mounted on a multiphoton microscope for simultaneous imaging under sustained uniaxial/biaxial loads.
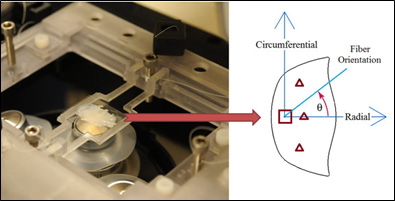
We have recently implemented this technique to study the load dependent EMC orientation of porcine atrioventricular (AV) heart valves. AV valves control the blood flow between the atria and ventricles and regurgitation is their most common symptom. Our data indicates that despite similarities in histological and immunohistochemical data acquired in our lab, the collagen fiber distribution and orientation in the stress-free condition were found to be different. Also, during the stress-free state, the fibers are scattered in a smaller angle in the tricuspid valve than in the mitral valve. Under uniaxial loading both valves rearranged differently with fibers reorienting in-line with the load throughout the depth of the mitral leaflet but only reorienting in-line with the load in deeper layers of the tricuspid leaflet. Biaxial loading elicited similar response in both valves with fibers rearranging in between the two principal axes of the stresses independent of their relaxed state configuration. These findings can help understand various structural and degenerative native valve diseases, develop tissue engineered valves, and improve durability of bioprosthetic heart valves.
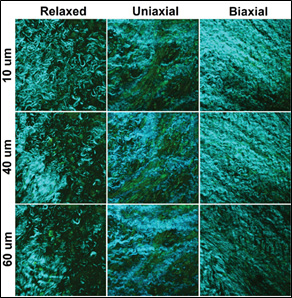
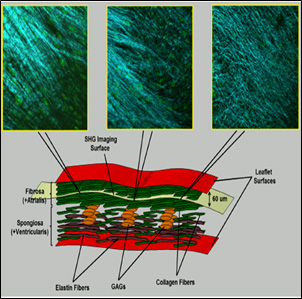
Schematic representation of a cross section of a heart valve leaflet shows the layered fashion of the tissue with all the extracellular matrix components.
Elastin (brown), collagen (green), and glycoaminoglycans (GAGs, orange) are distributed in the fibrosa and spongiosal layers. The fibrosa layer is rich in collagen fibers and more suitable for studying the load-dependent tissue remodeling. The Second Harmonic Generation images at the top were taken at the same depth (60 µm) from the surface of tissue inside the fibrosa. These images display different collagen density but almost similar fiber orientation o a plane paralel tothe surface of the leaflet.
View all research projects



